A New Chapter in Pain Medicine — With Important Caveats
Fifty million Americans live with chronic pain, and for many, current treatments fall short. Opioids carry addiction risks. Anticonvulsants help some but not others. NSAIDs have limits. For patients with persistent neuropathic pain — the burning, shooting, electric sensation that follows nerve damage — options have been particularly limited.
Until now, perhaps.
Over the last several months in our practice, we’ve prescribed suzetrigine for patients with various forms of neuropathic pain. Suzetrigine is currently approved only for acute pain, but its mechanism as a selective sodium channel blocker suggests broader applications. From our limited experience, many patients — even those with years-long neuropathy — seem to respond, including patients with post-herpetic neuralgia and certain forms of post-surgical pain.
This is anecdotal experience from a single center, and these observations require careful clinical trials before drawing conclusions. But the early signals are encouraging in ways we haven’t seen for years.
An evolving therapeutic class
Suzetrigine (Journavx) became the first non-opioid analgesic approved by the FDA in over 20 years when it received approval on January 30, 2025. But it represents the culmination of years of research into selective NaV1.8 sodium channel blockers rather than a singular breakthrough. Earlier compounds in this class demonstrated the therapeutic potential but faced development challenges or limited clinical advancement.
Suzetrigine’s success validates the broader strategy of targeting NaV1.8, a voltage-gated sodium channel expressed selectively in peripheral nociceptive neurons, with >31,000-fold selectivity compared to other sodium channel subtypes. The precision matters clinically. Because NaV1.8 has minimal expression in the brain, suzetrigine provides analgesia without central nervous system effects, including addiction potential.
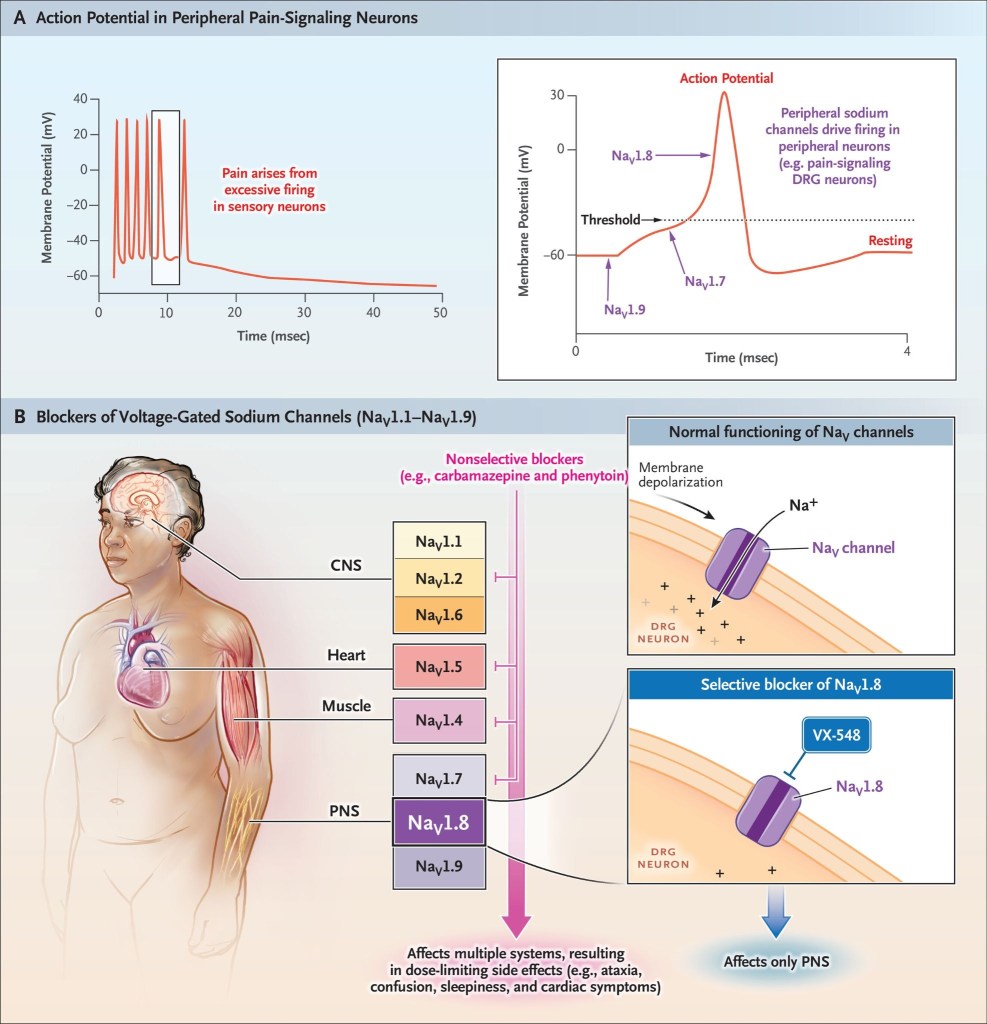
Waxman, S. G. (2023). Targeting a peripheral sodium channel to treat pain.
New England Journal of Medicine, 389(5), 466-469.
What distinguishes suzetrigine is its unique mechanism: potent state-dependent inhibition with “reverse use dependence.” It binds tightly to channels in their resting state and maintains tonic inhibition under physiological conditions, different from other sodium channel inhibitors.
In clinical trials, patients experienced statistically significant pain reduction compared to placebo in post-surgical settings, with common side effects limited to mild gastrointestinal symptoms, itching, and muscle spasms.
But we need to acknowledge the gaps
FDA approval was based on just two trials — abdominoplasty and bunionectomy — involving approximately 2,200 patients evaluated for 48 hours. That’s the entire foundation: two surgical procedures in relatively young, healthy patients.
We have zero long-term safety data for continuous use, no head-to-head comparisons with gabapentin or pregabalin, and no dedicated trials in the neuropathic pain conditions we’re now treating off-label. (Vertex Pharmaceuticals are now running a Phase 3, Open-label Study Evaluating the Long-term Safety and Effectiveness of Suzetrigine in Subjects With Pain Associated With Diabetic Peripheral Neuropathy).
The mechanistic assumptions aren’t ironclad either — NaV1.8 knockout studies showed mixed results, with some pain states proving NaV1.8-independent. The broader sodium channel field is littered with promising compounds that failed in humans despite strong preclinical rationale.
The road not taken: tetrodotoxin
The contrast with another sodium channel blocker is instructive. Tetrodotoxin, derived from pufferfish, has been in clinical trials since 2007 for cancer pain and neuropathy. A recent systematic review and meta-analysis of five clinical trials showed TTX significantly increased the number of patients achieving ≥30% pain improvement compared to placebo, with an effect size of 0.68 (p = 0.0065). More remarkably, it showed a median duration of 56.7 days of analgesia after just four days of treatment, compared to 9.9 days for placebo.
Yet across all completed trials, only 298 patients received the optimal 30 µg twice-daily dose — a vanishingly small evidence base for over 15 years of development.
Here’s where the story gets interesting: TTX is also one of the most lethal natural toxins known, about 1,000 times stronger than potassium cyanide. Just 10 grams of pufferfish liver contains enough TTX to kill a person. The Japanese health ministry reports a 2.8% fatality rate from pufferfish poisoning, with death occurring within four to six hours as paralysis spreads while consciousness remains clear until the very end. Not exactly your typical drug development starting point. (Here is a remarkable story of 12 y/o boy who became a certified expert in the art of fugu preparation).
Yet researchers have spent nearly two decades trying to harness this deadly compound for pain relief. It’s the classic pharmacological paradox — the difference between poison and medicine is often just the dose. Here’s the fascinating irony: TTX blocks most TTX-sensitive sodium channels, but both NaV1.5 (cardiac) and NaV1.8 (suzetrigine’s target) are TTX-resistant. Suzetrigine targets precisely what TTX can’t reach, while TTX blocks nearly everything else. The ancient neurotoxin required continuous monitoring for cardiac and respiratory effects; the precision-designed molecule can be taken as an outpatient oral medication.
The TTX program showed promising efficacy signals but faces financial uncertainty despite demonstrating safety (no TTX-related deaths across all studies, though increased non-severe adverse events). Suzetrigine succeeded where an ancient neurotoxin with impressive duration couldn’t quite reach approval.
It’s a perfect illustration of how modern drug development has evolved from “controlled poisoning” toward precision molecular targeting.
What comes next
Vertex is conducting Phase 3 trials in diabetic peripheral neuropathy and planning studies in lumbosacral radiculopathy. These will be the real test — dedicated studies in chronic neuropathic pain populations rather than acute post-surgical settings. However, early data from Phase 2 and 3 trials for chronic pain have shown mixed results, with clear efficacy for acute pain but unresolved questions regarding chronic neuropathic conditions.
I’m cautiously optimistic while acknowledging we’re working with incomplete data. The patients responding in our practice represent exactly the population that’s been underserved — those with persistent neuropathic pain who’ve exhausted conventional options.
But until controlled trials confirm what we’re seeing, we should be humble about what we know versus what we hope.
What we still need
Long-term safety data. Head-to-head comparisons with standard treatments. Dose optimization for chronic pain. Biomarkers to identify responders.
There’s also the practical reality of access. Suzetrigine carries a significant cost, and insurance coverage remains limited. The manufacturer offers a patient assistance program, but it’s time-limited and has eligibility restrictions. This creates an uncomfortable scenario: patients may experience relief during the coupon period only to face discontinuation due to cost. It’s a reminder that even promising new therapies must navigate the broader healthcare access landscape.
The current off-label use fills a clinical need but shouldn’t be confused with evidence-based practice.
Still, when patients with years of neuropathy report meaningful relief from a non-addictive oral medication, that matters. We haven’t had a new tool like this in decades. The mechanism is sound, the early signals are promising, and the unmet need is enormous.
Sometimes progress comes through unexpected paths — precision engineering succeeding where ancient toxins couldn’t quite reach approval. If the ongoing trials confirm our clinical observations, sodium channel blockers could reshape neuropathic pain management. Until then, careful monitoring and honest acknowledgment of what we don’t yet know.
What’s your experience been? If you’re treating patients with chronic neuropathic pain, what tools are working? What gaps do you see in current management? I’d love to hear your thoughts — reply to this newsletter or connect with me on LinkedIn.
Disclosure: I have no financial relationships with Vertex Pharmaceuticals, WEX Pharmaceuticals, or any conflicts of interest related to suzetrigine or tetrodotoxin.
